

Characterization Of Cobalt Nanoparticle Obtained from Green Synthesis and Its
X-Ray Diffraction Analysis
Ruby Baruah
Department of Chemistry, Tinsukia College, Tinsukia, Assam
Corresponding Author: rubytsk2020@gmail.com
Abstract
Green synthesis of nanoparticles is an eco-friendly process in which biological sources like leaves or plant extracts. This process can be used to synthesize cobalt nanoparticles (Co3O4). In this research work CoNPs were synthesized with Guava and Pomelo leaf extract. Characterization of CoNPs was carried out by taking FTIR and X-ray Diffraction analysis. Both samples showed Co-O bonds in the 600–400 cm−1 range, indicating cobalt oxide formation. The X-ray diffraction analysis also showed that there is no metallic cobalt instead there are mixed CoO and Co3O4 particles. The particle sizes were calculated from the X-ray diffraction analysis of the cobalt nano particle by using Scherrer equation and were found to be 66.2 nm and 85.5nm.
Key Words: CoNPs, leaf extract, green synthesis, Scherrer equation, Spectra.
Introduction
CoNPs have broad-spectrum antimicrobial activity against bacteria, viruses, and fungi which disrupt microbial cell membranes, interfere with enzyme function, and damage DNA. Cobalt nanoparticles (CoNPs) are particles of cobalt with dimensions in the range 1-100 nm. These nanoparticles have unique physical, chemical, and biological properties that distinguish them from bulk cobalt, making them highly valuable in a variety of applications. The exact SPR (surface plasmon resonance) peak position depends on the size, shape, and dielectric environment of the nanoparticles. Cobalt nanoparticles can also act as catalysts in various chemical reactions, including the reduction of organic dyes and the oxidation of carbon monoxide. CoNPs are also used in wound dressings for their antimicrobial properties. They are also used as coatings on medical devices to prevent infections. While CoNPs have numerous beneficial applications, concerns about their potential toxicity and environmental impact have been raised. Cobalt nanoparticles can be toxic to aquatic organisms and may pose health risks to humans if not properly managed.
Research is ongoing to understand the long-term effects of CoNPs and to develop safe handling, disposal, and regulatory methods (G. Asha et al.,2022). Nano-sized materials are synthesized in a different way, but there are generally two approaches to their creation: top-down and bottom-up synthesis. The former utilizes larger bulk material and breaks them down into nano-sized particles, and the latter utilizes individual atoms and builds them up into larger nanomaterials.
There are different methods for nano synthesis. Green synthesis is needed to create materials and chemicals in a more sustainable and environmentally friendly way. It aims to reduce or eliminate the use of harmful chemicals and materials, minimize waste generation, and promote the use of renewable resources and energy. Green synthesis methods often use non- toxic and biodegradable materials and tend to generate less waste and pollution. These methods can be more cost-effective than physical and chemical methods; often utilize renewable resources, such as plant extracts.
Synthesis of Cobalt Nanoparticles by Chemical approaches
Chemical approaches for synthesizing cobalt nanoparticles involve the reduction of cobalt ions to metallic cobalt using chemical agents. These methods typically offer precise control over the size, shape, and properties of the nanoparticles. Common chemical approaches for CoNPs synthesis:
Reduction of cobalt salts by Sodium Borohydride (NaBH4)
Cobaltous sulphate is mixed with a reducing agent such as sodium borohydride (NaBH4) in an aqueous solution. The reducing agent (NaBH4) donates electrons to the cobalt ions, leading to their reduction to metallic cobalt and the formation of CoNPs. The reaction is typically carried out under controlled conditions of temperature, pH, and reaction time to regulate the size and morphology of the nanoparticles.
Reduction by Citrate Method
This method involves the reduction of cobalt ions by citrate ions (e.g., sodium citrate) in an aqueous solution. Cobaltous sulphate is mixed with a citrate solution and heated to promote the reduction reaction. The citrate ions act as both reducing agents and stabilizing agents, preventing the aggregation of nanoparticles and controlling their size distribution.
Reduction by Polyol Process
In the polyol process, a polyol solvent such as ethylene glycol or glycerol is used as both a solvent and a reducing agent. Cobaltus sulphate is dissolved in the polyol solvent, and a reducing agent such as polyvinylpyrrolidone (PVP) is added to facilitate the reduction of silver ions. The reaction is typically carried out at elevated temperatures under inert atmosphere conditions to promote the formation of uniform nanoparticles.
Solvothermal Synthesis
High-temperature synthesis: Synthesizing cobalt nanoparticles in a solvent at high temperatures and pressures.
These chemical approaches offer versatility and scalability for the synthesis of cobalt nanoparticles with tailored properties for various applications in catalysis, electronics, sensing, and biomedical fields. However, it’s important to optimize reaction conditions and characterize the synthesized nanoparticles to ensure their uniformity, stability, and desired properties.
Synthesis of Cobalt Nanoparticles by Biological approaches
Synthesis of cobalt nanoparticles (CoNPs) using biological approaches, also known as green synthesis, involves the use of natural extracts, microorganisms, or other biological entities to reduce cobalt ions to cobalt nanoparticles. These methods are preferred over conventional chemical methods due to their eco-friendliness, cost-effectiveness, and potential for large-scale production.
Some common biological approaches for synthesizing cobalt nanoparticles: Various plant extracts contain bioactive compounds such as flavonoids, phenolics, and terpenoids that can act as reducing and stabilizing agents for cobalt nanoparticles. Plant extracts are mixed with aqueous cobaltous sulphate solution and heated or exposed to sunlight to facilitate the reduction process. Moringa oleifera, neem, aloe vera, tulsi (holy basil) etc are some plants whose extract can be used for the synthesis of CoNPs.
Certain microorganisms, including bacteria, fungi, and algae, possess enzymes or biomolecules that can reduce cobalt ions to CoNPs. Bacterial species like Escherichia coli, Bacillus subtilis, and Pseudomonas aeruginosa, as well as fungal species like Aspergillus, Fusarium, and Candida, have been used for this purpose. Microbial biomass is typically incubated with cobalt sulphate solution under suitable conditions (e.g., temperature, pH) to induce nanoparticle formation.
Algae and cyanobacteria are capable of synthesizing CoNPs due to the presence of bioactive compounds like polysaccharides, proteins, and pigments. These microorganisms are cultured in the presence of cobalt ions, and the reduction of cobalt ions to nanoparticles occurs intracellularly or extracellularly. Marine algae and cyanobacteria are particularly promising sources for CoNPs synthesis.
Enzymes such as nitrate reductase, alcohol dehydrogenase, and cytochrome c can catalyse the reduction of silver ions to CoNPs. Enzymatic synthesis offers high specificity and control over the nanoparticle size and shape. Enzymes are isolated from biological sources or produced recombinantly and mixed with cobalt ion solutions to initiate nanoparticle formation.
Biological fluids such as blood plasma, urine, and plant sap contain a variety of biomolecules (e.g., proteins, sugars, metabolites) that can act as reducing and stabilizing agents for CoNPs. Cobaltous sulphate solution is mixed with biological fluids under controlled conditions to induce nanoparticle formation. Biological approaches for synthesizing cobalt nanoparticle offer several advantages, including biocompatibility, scalability, and reduced environmental impact. Moreover, the resulting nanoparticles often exhibit enhanced stability and biocompatibility, making them suitable for various applications in biomedicine, catalysis, electronics, and environmental remediation (J.K. Patra et al., 2014).
Materials and Methods
Collection of Plant Samples
The leaf of Psidium guajava (Guaya) and Citrus maxima (Pomelo) plant was collected. Guaya leaves are often used to treat stomach pain, diarrhoea and Pomelo leaves are used to treat coughs, respiratory discomfort.
Fig: 1: Guava & Pomelo Leaves
Preparation of Plant Extract
Fresh and healthy leaves of guava and pomelo were collected and rinsed with tap water followed by deionised water and allowed to air dry. Once the leave got dried, they were cut into small pieces. After that, about 10 grams of it was taken in a 500ml beaker containing 100ml distilled water and was refluxed for 2 hours. After cooling, they were filtered by using filter paper. The extract obtained is used for further process.
Preparation of Cobaltous Sulphate
0.5M solution of Cobaltous sulphate was prepared by accurately weighing 14.06 g of cobaltous sulphate (AR, Merck) heptahydrate in 100ml of distilled water. The solution was prepared in coloured bottle and kept in dark as it reacts in presence of light.
Synthesis of CoNPs Using Plant extract
About 10ml of plant extract which was prepared, was taken in a 100 ml conical flask and 10 ml of 0.5M CoSO4 solution was added to it and kept at room temperature for reduction and the change of colour was monitored, which indicates the formation of nanoparticles. The separation of purification of nanoparticles was done (H. Ali et al and M. Abdi et al,2023).
FT-IR Analysis
FTIR of the cobalt samples were taken in Dibrugarh University, Dibrugarh, Assam, India with PerkinElmer Spectrum Version 10.7.2.
X-ray Diffraction Analysis
The X-ray Diffraction analysis of the sample was done in Central Sophisticated Instrumentation Centre, Dibrugarh University, Dibrugarh, 786004. The Scherrer equation is
τ = Kλ / βcos θ
Where τ is the size of the particle. K is a dimensionless shape factor, with a value close to unity, β is the line broadening at half the maximum intensity, θ is the Bragg’s angle.
Results and Discussion
Characterization Of Cobalt Nanoparticles (Yu A. Mendoza-Garcia et al., 2013 and S. Rana et al., 2010)
The Fourier transform infrared (FTIR) method is a type of spectroscopy that can detect changes in the total composition of biomolecules by determining changes in functional groups. The vibration and rotation of molecules influenced by infrared radiation at a particular wavelength is measured using FTIR. The basic principle of FTIR spectroscopy is that molecules absorb infrared radiation at specific frequencies that correspond to the vibrational modes of their bonds. When IR light passes through or reflects off a sample, certain wavelengths are absorbed, and the resulting spectrum shows absorption peaks at those frequencies.
FTIR Spectrum of CoNPs synthesized from Guava leaf extract
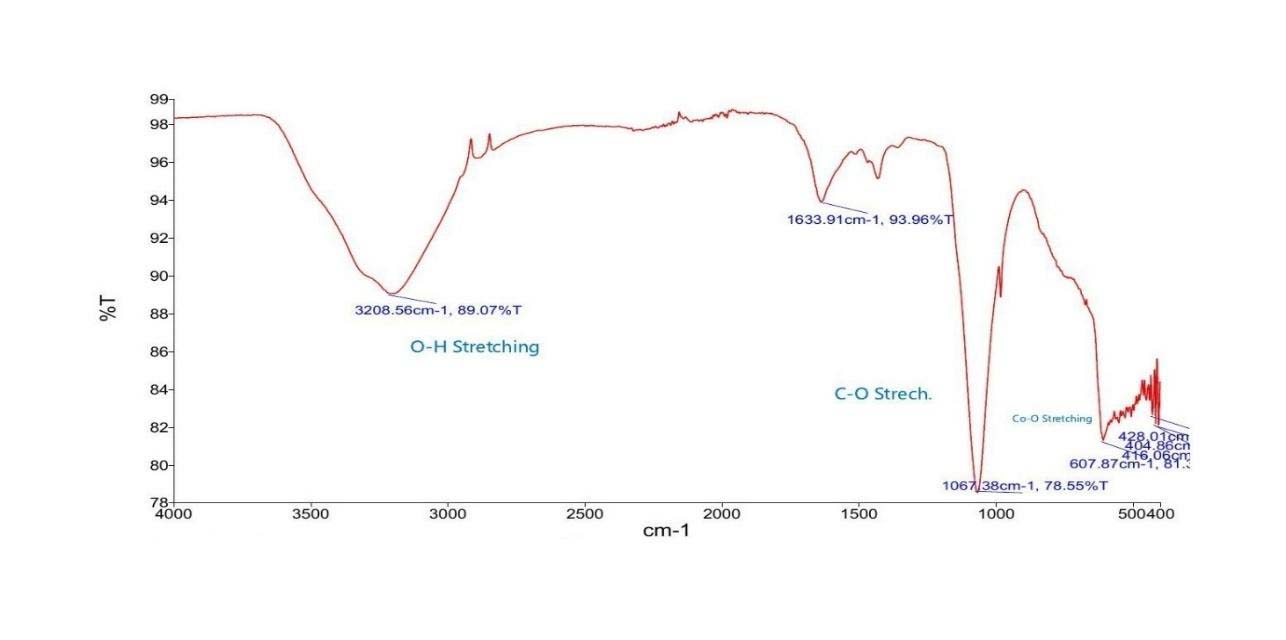
Key Peaks Observed in CoNPs obtained from guava leaf
- 3208 cm−1 (O–H stretch): From hydroxyl-rich compounds such as quercetin, ellagic acid, and flavonoids in guava.
- 1633 cm−1 (C=C or C=O stretch): Likely from aromatic rings or carbonyls in polyphenols or tannins.
- 1067 cm−1 (C–O stretch): Indicative of phenolic or glycosidic linkages.
- 607–428 cm−1 (Co–O vibrations): Confirms formation of cobalt oxide (Co3O4 or CoO).
Summary of Functional Groups Likely Present
- Hydroxyl group (–OH) – broad peak at ~3208 cm−1.
- Possibly C=C or amide-related bending – peak around 1634 cm−1.
- C–O stretching – strong peak at ~1067 cm−1.
- Inorganic or aromatic ring structures peaks below 700 cm−1.
Guava extract provides strong reducing and capping agents, as shown by intense organic functional group signals. These nanoparticles are well-capped and potentially stable.
FTIR Spectrum of CoNPs Synthesised from Pomelo Leaf Extract
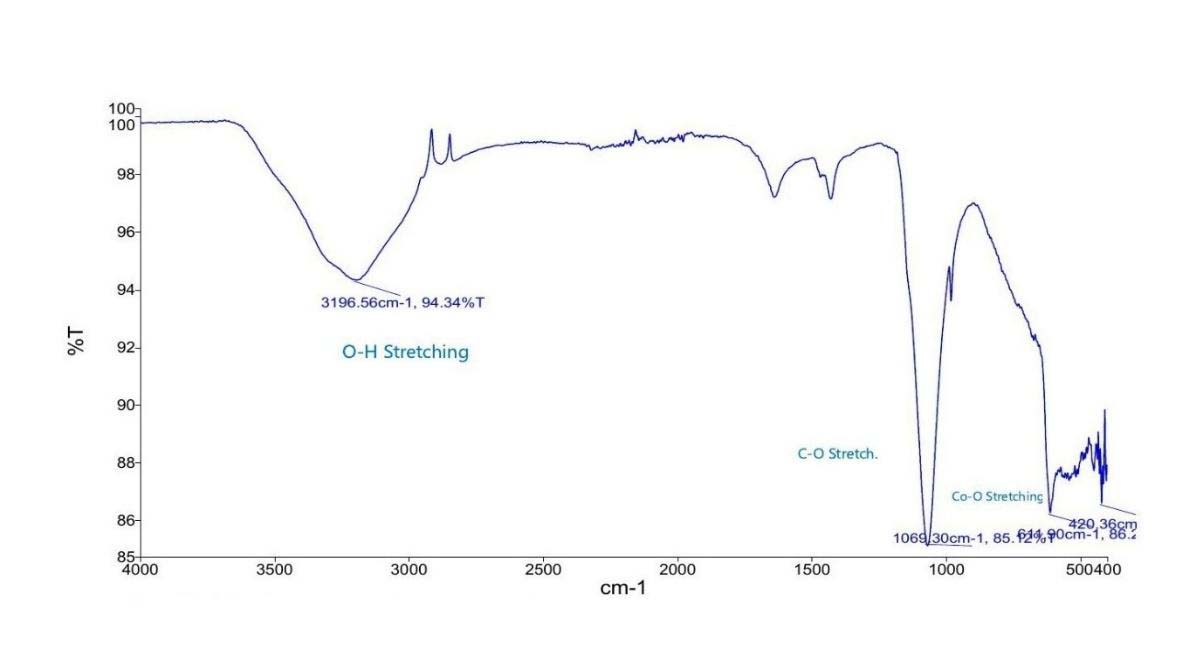
Key Peaks Observed
- 3196 cm−1 (O–H stretch): From flavonoids or essential oils like naringin in pomelo.
- Weak/absent 1600 cm−1 peak: Indicates less aromatic or carbonyl residue, possibly due to lower concentration or better washing.
- 1065 cm−1 (C–O stretch): Still present, confirming organic involvement.
- 614–429 cm−1 (Co–O): Clear evidence of cobalt oxide nanoparticle formation.
- Pomelo-synthesized nanoparticles show less capping or organic residue, which may mean cleaner surfaces, potentially offering different reactivity or catalytic behaviour.
X-ray Diffraction Analysis (Giriraj Tailor et al,2025 and Dhaneswar Das et al.,2023)
From the XRD analysis of the sample the phase identification can be done. The metallic cobalt exists as hcp (α-Co) and fcc (β-Co) and forms (CoO, Co3O4) have distinct peaks. The characteristics peaks can be shown as follows:
Table1: Characteristics peaks for hcp (α-Co) and fcc (β-Co)
| hcp(α-Co) | (approx..) 2θ |
| (100) | (approx..) 41.56° |
| (002) | (approx..) 44.50° |
| (101) | (approx..) 47.42° |
| Fcc(β-Co) | (approx..) 2θ |
| (111) | (approx..) 44.22° |
| (200) | (approx..) 51.52° |
| (220) | (approx..) 75.84° |
Table.2: The peak list of XRD of the cobalt sample.
| SL. No. | 2-theta(deg) | d(ang.) | Height (counts) |
FWHM (deg) |
Phase name | Chemical formula |
DB card number |
|---|---|---|---|---|---|---|---|
| 1 | 15.16(3) | 5.838(10) | 381(20) | 0.32(8) | Unknown | Unknown | 0 |
| 2 | 16.402(8) | 5.400(3) | 929(30) | 0.20(4) | Unknown | Unknown | 0 |
| 3 | 16.852(14) | 5.257(4) | 327(18) | 0.07(4) | Unknown | Unknown | 0 |
| 4 | 18.13(14) | 4.89(4) | 464(22) | 2.50(14) | Unknown | Unknown | 00-000-0000 |
| 5 | 18.165(3) | 4.8798(8) | 7336(86) | 0.077(3) | Unknown | Unknown | 0 |
| 6 | 18.434(4) | 4.8090(10) | 3140(56) | 0.103(7) | Unknown | Unknown | 0 |
| 7 | 19.632(3) | 4.5182(7) | 1467(38) | 0.065(7) | Unknown | Unknown | 0 |
| 8 | 20.265(9) | 4.3785(19) | 2587(51) | 0.115(17) | Unknown | Unknown | 0 |
| 9 | 21.33(10) | 4.1610(19) | 1531(39) | 0.23(2) | Unknown | Unknown | 0 |
| 10 | 22.103(7) | 4.0184(13) | 2395(49) | 0.080(10) | Unknown | Unknown | 0 |
| 11 | 23.6528(18) | 3.7584(3) | 7587(87) | 0.073(3) | Unknown | Unknown | 0 |
| 12 | 23.978(5) | 3.7082(7) | 1966(45) | 0.047(14) | Unknown | Unknown | 0 |
| 13 | 24.75(9) | 3.594(12) | 366(19) | 2.0(2) | Unknown | Unknown | 0 |
| 14 | 27.467(4) | 3.2446(5) | 2817(53) | 0.063(5) | Unknown | Unknown | 0 |
| 15 | 28.104(9) | 3.1725(10) | 1438(38) | 0.093(8) | Unknown | Unknown | 0 |
| 16 | 28.588(6) | 3.1198(6) | 455(21) | 0.054(19) | Unknown | Unknown | 0 |
| 17 | 28.858(6) | 3.0912(7) | 654(26) | 0.13(2) | Unknown | Unknown | 0 |
| 18 | 29.269(10) | 3.0487(10) | 531(23) | 0.25(4) | Unknown | Unknown | 0 |
| 19 | 29.742(6) | 3.0014(6) | 778(28) | 0.24(3) | Unknown | Unknown | 0 |
| 20 | 30.58(5) | 2.921(4) | 620(25) | 0.26(8) | Unknown | Unknown | 0 |
| 21 | 31.870(18) | 2.8056(15) | 236(15) | 0.08(5) | Unknown | Unknown | 0 |
| 22 | 32.140(5) | 2.7827(4) | 1069(33) | 0.097(13) | Unknown | Unknown | 0 |
| 23 | 32.869(5) | 2.7226(4) | 2229(47) | 0.076(7) | Unknown | Unknown | 0 |
| 24 | 33.373(5) | 2.6827(4) | 337(18) | 0.048(19) | Unknown | Unknown | 0 |
| 25 | 33.987(5) | 2.6356(4) | 1533(39) | 0.067(6) | Unknown | Unknown | 0 |
| 26 | 34.466(3) | 2.6000(2) | 1083(33) | 0.062(9) | Unknown | Unknown | 0 |
| 27 | 36.391(10) | 2.4668(6) | 928(30) | 0.079(9) | Unknown | Unknown | 0 |
| 28 | 36.85(2) | 2.4374(16) | 357(19) | 0.08(2) | Unknown | Unknown | 0 |
| 29 | 37.32(2) | 2.4075(16) | 611(25) | 0.067(19) | Unknown | Unknown | 0 |
| 30 | 43.546(9) | 2.0766(4) | 389(20) | 0.10(3) | Unknown | Unknown | 0 |
| 31 | 45.081(11) | 2.0094(5) | 580(24) | 0.13(3) | Unknown | Unknown | 0 |
| 32 | 46.305(13) | 1.9591(5) | 900(30) | 0.078(10) | Unknown | Unknown | 0 |
| 33 | 48.37(2) | 1.8802(8) | 502(22) | 0.10(2) | Unknown | Unknown | 0 |
| 34 | 48.66(2) | 1.8697(9) | 209(14) | 0.07(4) | Unknown | Unknown | 0 |
| 35 | 49.071(6) | 1.8550(2) | 738(27) | 0.106(14) | Unknown | Unknown | 0 |
| 36 | 49.329(16) | 1.8459(6) | 355(19) | 0.13(4) | Unknown | Unknown | 0 |
| 37 | 49.614(10) | 1.8359(3) | 269(16) | 0.05(3) | Unknown | Unknown | 0 |
| 38 | 53.93(5) | 1.6987(15) | 198(14) | 0.08(6) | Unknown | Unknown | 0 |
| 39 | 56.658(11) | 1.6232(9) | 269(16) | 0.07(3) | Unknown | Unknown | 0 |
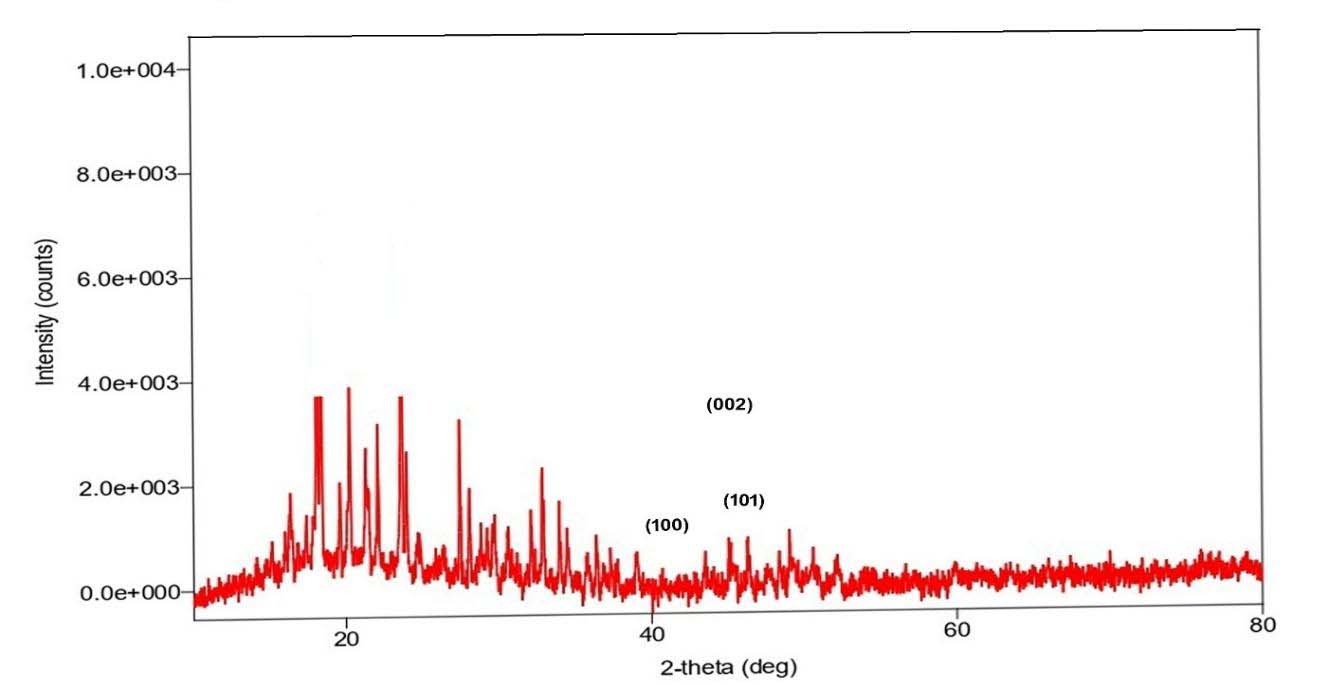
From the XRD values the strong peaks that match typical Co3O4 and CoO reflections were selected for particle size calculation using Scherrer equation (Sobra Nasiri et. Al,2023, and S.A. Hassanzadeh-Tabrizi, 2023). The results are shown in the Table.3
Table.3: Particle size of the sample (K = 0.9, λ = 1.5406 Å)
| 2θ (deg) | FWHM (deg) | D (nm) |
|---|---|---|
| 31.87 | 0.080 | 103.3 nm |
| 36.39 | 0.0799 | 104.7 nm |
| 43.55 | 0.100 | 85.5 nm |
| 45.08 | 0.130 | 66.2 nm |
| 56.66 | 0.070 | 128.9 nm |
Conclusion
FTIR analysis confirmed the successful synthesis of cobalt oxide nanoparticles using guava (Sample 1) and pomelo (Sample 2) leaf extracts. Both samples showed Co–O bonds in the 600–400 cm−1 range, indicating cobalt oxide formation. Guava-synthesized nanoparticles had more organic functional groups, suggesting stronger capping by phytochemicals. In contrast, pomelo-based nanoparticles showed fewer organic residues, indicating cleaner surfaces. This highlights that both plant extracts are effective, with guava offering better stability and pomelo potentially better reactivity.
From the X-ray Diffraction analysis, it is clearly seen that there is no metallic cobalt formed in this synthesis. The mixed cobalt oxide and Co3O4 is formed. However, the particle sizes of the cobalt oxides were found to be 66.2 nm and 85.5 nm. To obtain the metallic cobalt the thermal reduction should be done at 350-450°C as well as surfactants can be used to limit crystal growth.
Synthesis of fernet nanoparticles using plant extract is beneficial as it is economical, energy efficient and environment friendly. This green synthesis method can overcome other chemical and biological methods which involve various harmful chemical reagents as well as time consuming process of employing microbes and maintaining their cultures.
Acknowledgement
Sincere gratitude to Dr. Bhairav Borgohain, Principal, Tinsukia College and Dr. Saswati Phookan, Head of the department, Department of Chemistry, Tinsukia College, Tinsukia, Assam for their constant support and providing the necessary facilities to carry out this research. I am thankful to Mr. Karunakor Borah, Technical officer, CSIC, Dibrugarh University, Assam for recording FTIR and XRD spectra for this research work.
Declaration
Conflict of interest: The author declare that they have no conflict of interest.
References
- Abdi, M., Yusuf, Z. & Sasikumar, J. M. (2024). Phyto-fabrication of cobalt oxide nanoparticles from Ocimum gratissimum L. leaf and flower extracts and their antimicrobial activities. Open Biotechnol. J. 17.
- Ali, H., Yadav, Y. K., Ali, D., Kumar, G. & Alarifi, S. (2023). Biosynthesis and characterization of cobalt nanoparticles using combination of different plants and their antimicrobial activity. Bioscience Reports 43.
- Asha, G., Rajeswari V., Stephen G., Gurusamy S. & Carolin Jeniba Rachel D. (2022). Eco-friendly synthesis and characterization of cobalt oxide nanoparticles by Satyum species and its photo-catalytic activity. Materials Today: Proceedings 48:486–493.
- Das D., Bhaskor & J Saikia J. (2023). Synthesis Characterization and Biological Application of Cobalt oxide (Co3O4) nanoparticles, Chemical Physics Impact, Vol.6.
- Hassanzadeh-Tabrizi S. A. (2023). Precise calculation of Crystallite size of nanomaterials: A review, Journal of Alloys and Compounds, Volume 968.
- Mendoza-Garcia Yu, A., Ning, B., & Sun S. (2013). Cobalt substituted magnetite nanoparticles and their assembly into ferrimagnetic nanoparticle arrays, Advanced Materials, vol. 25, no. 22: pp. 3090–3094.
- Nasiri S.& Rabie M. (2023). Modified Scherrer Equation to calculate Size by XRD with high Accuracy, Examples Fe 203, TiO2 and V2O5, Nano Trends,3.
- Patra, J. K. & Baek, K.-H. (2014). Green nanobiotechnology: factors affecting synthesis and characterization techniques. Journal of Nanomaterials. 1–12.
- Rana S., Philip J., & Raj B. (2010). Micelle based synthesis of cobalt ferrite nanoparticles and their characterization using Fourier Transform Infrared Transmission Spectrometry and Thermogravimetry, Materials Chemistry and Physics, 124(1), :264–269.
- Tailor G., Choudhury J., Singh S., Verma D. & Joshi M. (2025). Comprehensive Characterization of Cobalt oxide nanoparticles using microscopic and biochemical assay; Next Research, Vol.2, Issue 1.
