

Assessing Macrophyte Composition and Dynamics in the Naljhara River, Barpeta District, Assam
1*Sadananda Nath and 2Rodali Talukdar
1*Department of Zoology, B.H. College, Howly, Barpeta-781316, Assam, India
2Department of Chemistry, B.H. College, Howly, Barpeta-781316 Assam, India
Corresponding address: nathsadaa@gmail.com
Abstract
Macrophytes, both aquatic and semi-aquatic and other hydrophilic terrestrial vegetation play a significant role in the proper maintenance of the riverine ecosystem. Expansion and proliferation of these macrophytes at different seasons influence the proper continuation and distribution of biodiversity. The North Eastern region of India is considered one of the hot spots of freshwater biodiversity in the world, and the region has exceptional possibilities in the field of fisheries resources. However, these freshwater ecosystems are under tremendous pressure from various anthropogenic factors. The river Naljhara attains importance for its direct linkage with the rich aquatic resources of the Manas-Beki river system. The present study is an attempt to investigate the composition of macrophytes in the river Naljhara to make an assessment of the health of this freshwater ecosystem from a biodiversity point of view. The study was conducted during the year 2022-2023 in four different seasons across four different sampling sites using standard procedures. The study demonstrated that the river Naljhara in Barpeta district of Assam is represented by two groups of aquatic macrophytes, viz. Angiosperms and Pteridophytes. Both groups include a total of 21 families, under which there are 31 genera and 36 species. Among the four types of ecological categories of aquatic macrophytes, the rooted emergent or marginal category with 66.67 % of species composition is of highest percentage.
Keywords: Macrophytes, Naljhara River, freshwater ecosystem, biodiversity
Introduction
Macrophytes play an important role in controlling biotic structure, especially the dynamics of zooplankton, phytoplankton and fish population. They decrease the amount of nutrients available to phytoplankton but provide food and shelter to the zooplankton. Macrophytes serve as refuges or foraging ground and nursery areas to most fish species and serve as substratum for egg attachment and diminishing egg mortality (UNEP, 2004). Although some major secondary producers such as Cladocerans, rotifers and insect larvae are found both in pelagic regions and in the areas of vegetation but some of them are found only in or near the surrounding areas of stands of vegetation (Campbell et al., 1982).
Changes in the composition and abundance of macrophyte communities express the possible reasons behind any major changes in aquatic ecosystems, such as phenomena like eutrophication leads to changes in species composition and diversity. Therefore, macrophytes have gained importance for their role in detecting changes and are used in monitoring water quality (NERC, 1999). Macrophytes have low seasonal variability, and their community structure also changes slowly. Therefore, when a change is detected in macrophyte species composition, it indicates a probable change in the riverine ecosystem (Johson, 2001). Macrophytes release and utilise inorganic nutrients and dissolved organic compounds (Wetzel & Allen, 1970). Generally, aquatic macrophytes are classified as four morphotypes, i.e. submerged, floating-leaved, emergent and free-floating (Sculthorpe, 1985). Submerged macrophytes are the sites of food production, shelter and a favourable breeding ground for many aquatic animals and also act as substrata for attachment of periphyton (Diehl, 1992; Lodge, 1991; Rennie & Jackson, 2005). Submerged macrophyte communities, by providing habitat complexity, regulate abundance, diversity and distribution pattern of aquatic animals (Williams, 1980; Wilzbach, 1985).
In India, considerable studies were done on macrophyte diversity in the lentic freshwater bodies (Dey, 1981; Dey & Kar,1989; Kar & Barbhuiya, 2001; Sarma & Deka, 2014; Hazarika and Borthakur, 2014; Chan et al., 2022) but in lotic system, such studies are limited (Shah and Abbas, 1979; Pradhan et al., 2005; Dutta & Sarma, 2012). The North Eastern region of India is considered one of the hot spots of freshwater biodiversity in the world, and the region has exceptional possibilities in the field of fisheries resources. However, these freshwater ecosystems are under tremendous pressure from various factors such as rapid urbanization, deforestation and shrinking of forest cover, embankment of rivers, extraction of river water for irrigation, siltation of river beds, industrial and domestic water pollution, large scale use of insecticides in the agricultural field, over exploitation of fishes using small mesh size nets, chemicals, poisons and explosives. The river Naljhara attains importance for its direct linkage with the rich aquatic resources of the Manas-Beki river system. The river Naljhara carries huge sediment load and get subjected to flash flood and frequent shifting of channel during flood. The river Naljhara since flows through an agricultural catchment, it faces numerous challenges. The present study is an attempt to investigate the composition of macrophytes in the river Naljhara to make an assessment of the health of this freshwater ecosystem from a biodiversity point of view.
Materials and Methods
Study area
The river Naljhara flows through the district of Baksa and Barpeta as a separate channel and joins with the river Beki within the Barpeta district of Assam. The River Naljhara is a split channel of the river Manas. It is situated between 26°31’28.97” North to 26°33’39.57” North latitude and 90°53’3.34” East to 90°54’41.90” East longitude in the Chakchacka and Gobardhana Development Block under the Barpeta district of Assam. The river Naljhara forms a major connecting network between the river Manas and the river Beki. The river represents a part of the northern tributary of the Brahmaputra River. The river Naljhara traverses about 13-15 km within the Barpeta district of Assam. The climate of the study area is warm and humid. Summer seasons are hot, followed by monsoon of heavy rainfall, and winter seasons are relatively cool and dry. During the period from October to March, periodic dry spells occur. Heaviest rainfall occurs during the period from May to July.
Study Design
The study was conducted from March 2022 to February 2023 in four different seasons as per the seasons described by Borthakur (1986). The seasons are –pre-monsoon (March-May; Identity-PM); monsoon (June-September; Identity-M), retreating monsoon (October-November; Identity-RM), and winter (December-February; Identity-W). Four different sampling sites had been selected on random selection starting from the confluence point with the Beki river. Three replicate sampling was done in each season at four different sampling sites. The details of sampling sites are shown in Table 1.1 and Map. Aquatic macrophytes were collected by hand and with the help of a knife and packed in plastic bags for making dry herbarium following usual laboratory techniques and some macrophytes were also kept in bottles filled with 70% formalin acetone alcohol (Jain and Rao, 1977). Specimens were identified up to the lowest possible taxonomic level by using key factors given in standard literature (Hutchinson, 1975; Biswas & Calder, 1984; Fassett, 1997). The ecological categorization of macrophytes was done as per the description of Sculthorpe (1985). The past software programme was used for statistical analysis and estimation of the Bray-Curtis Similarity index.
| Sampling site | Name | GPS location |
|---|---|---|
| S1 | Jamadarbari Pathar | 26°31’28.97” N and 90°54’41.90” E |
| S2 | Khudnabari Pathar | 26°31’54.78” N and 90°54’25.54” E |
| S3 | Kanar Gaon | 26°32’18.73” N and 90°53’41.74” E |
| S4 | Unnekuri | 26°33’9.57” N and 90°53’3.34” E |
Results and Discussion
Composition of Macrophytes
In the present investigation, the observed and recorded macrophyte community from Naljhara River in Barpeta district of Assam is represented by 36 species of 31 genera, 21 families and 02 groups. The maximum numbers of macrophyte species (7 species) were represented by the family Poaceae with 19.44% of species composition, followed by the family Cyperaceae (5 species) with 13.89 % of species composition. The family Hydrocharitaceae with 3 nos. of species and family Amaranthaceae, Pontederiaceae, and Potamogetonaceae with 2 nos. of species each exhibit 8.33% and 5.56% of species composition, respectively. All other families, such as Alismataceae, Aponogetonaceae, Araceae, Asteraceae, Ceratophyllaceae, Commelinaceae, Lemnaceae, Lentibulariaceae, Menyanthaceae, Onagraceae, Plantaginaceae, Polygonaceae, Typhaceae, Azollaceae and Marsileaceae, were represented by only 1 species, with 2.78% of species composition. Among the two identified groups of aquatic macrophytes, the majority of species were represented by the Angiosperms and the group Pteridophyta was represented by only 2 species, i.e. Azolla filiculoides Lam. under family Azollaceae and Marsilea quadrifolia L. under family Marsileaceae. The details are shown in Table 1.2; Fig. 1.2 & Fig. 1.3.
Ecological Categories of Macrophytes
Based on the adaptation of macrophytes with the existing conditions of the Naljhara river, 4 ecological categories of the macrophyte element were identified. The Ecological classification of aquatic macrophytes of Naljhara river exhibits, higher percentage of macrophytes under the rooted emergent or marginal category, with 66.67 % of species composition. The other categories, such as Free floating, rooted emergent with floating leaves and rooted submerged, were represented by 11.11% of species composition. Free-floating macrophytes include 4 species, such as Azolla filiculoides Lam., Eichhornia crassipes (Mart.) Solms., Lemna perpusilla Torr. and Utricularia aurea Lour.; the Rooted emergent with floating leaves includes 4 species, such as Aponogeton sp., Hydrocharis sp., Potamogeton nodosus Poir and Sagittaria guayanensis Kunth; and rooted submerged includes 4 species, such as Blyxa aubertii Rich, Ceratophyllum sp., Hydrilla sp., and Potamogeton crispus L. The other remaining macrophytes were categorized under the Rooted emergent or marginal class, with the majority of species from the families Cyperaceae and Poaceae. The details are shown in Table 1.3 & Fig. 1.4.
Distribution and Diversity of Macrophytes
The distribution status of aquatic macrophytes exhibits a heterogeneous picture in the selected sampling sites of the Naljhara River. During the study period, no characteristic change was detected in the species richness of macrophytes of the Naljhara river at the selected sampling sites in different seasons. The maximum number of 33 species of macrophytes was observed during the winter season at the sampling sites S1 and S2. Similarly, at the sampling sites S3 and S4, the maximum number of 15 and 13 species of macrophytes was observed in the winter seasons, respectively. The minimum number of 1 species of macrophyte was observed at all sampling sites during the monsoon season. The details are shown in Table 1.4 & Table 1.5.
Cluster Analysis of Sampling Sites
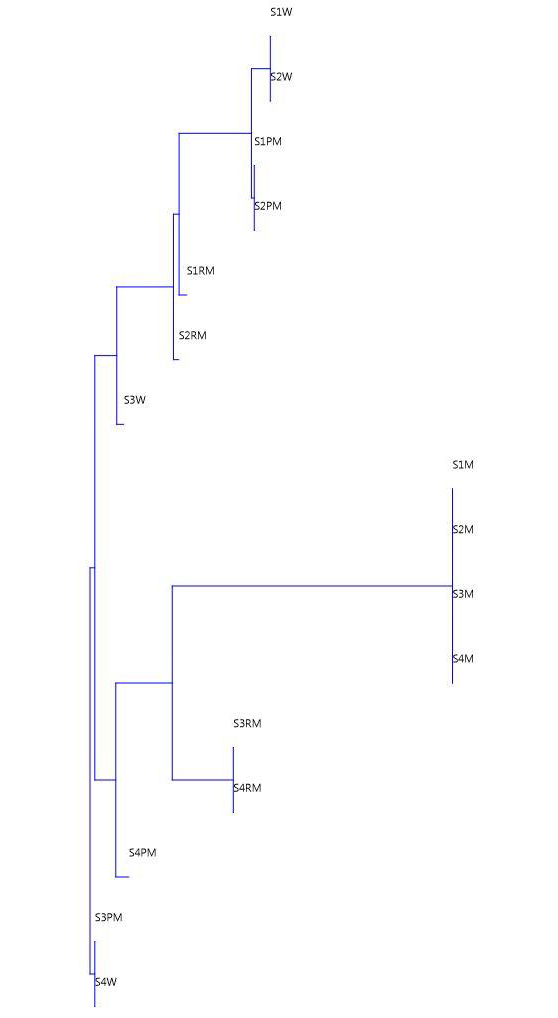
Cluster analysis through Bray-Curtis similarity index of the sampling sites based on the distribution status of the number of Species (Taxa) of macrophytes at the sampling sites of Naljhara river in different seasons shows a comparable picture with neighbouring sampling sites joining clustering. The details are shown in Fig. 1.5. Monsoon season of the sampling sites number S1, S2, S3 and S4 have joined a cluster due to the presence of only 1 number of species. Similarly, the winter seasons of S1 and S2 have joined clustering due to the presence of 33 numbers of species. Joining of clustering was observed between Sand S2 in the pre-monsoon season with 30 species, S3 and S4 in the retreating monsoon with 7 species, S3 in the pre-monsoon season and S4 in the winter season with 13 species, S1 and S2 in the retreating monsoon season with 21 and 20 species respectively. In the dendrogram, sampling site number S3 in the winter season and S4 in the pre-monsoon season was identified as out-group, without joining clustering due to the presence of 15 and 11 species, respectively. The presence of a limited number of macrophytes during the monsoon season in all the sampling sites in the present study is associated with the prevalence of high-water depth and flow during the monsoon season in comparison to the other seasons. The present investigation demonstrates that shallow water with less water current during the pre-monsoon, retreating monsoon and winter seasons is ideal habitat for the growth of macrophytes and with increase in depth and water current during monsoon season macrophyte composition and diversity decreases. These findings are in agreement with the work of Riis et al., (2019) and Rukl et al., (2020).
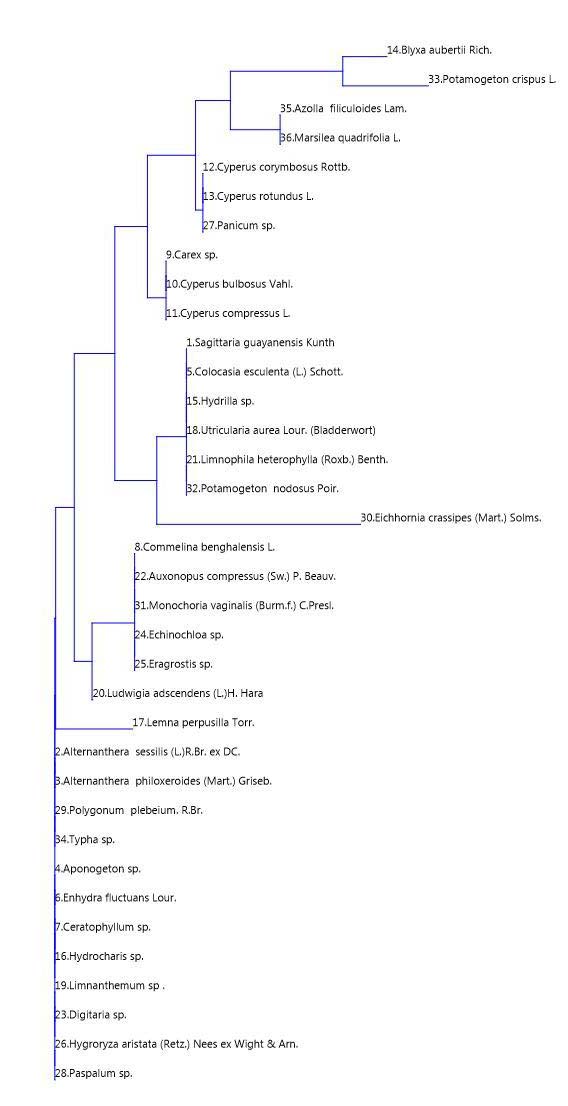
Cluster Analysis of Macrophyte Species
Cluster analysis through Bray-Curtis similarity index of the macrophyte species based on the distribution status of macrophytes of Naljhara river at selected sampling sites in different seasons shows a comparable picture with neighbour macrophytes joining clustering. The details are shown in Fig. 1.6. Joining of clustering was observed between Potamogeton crispus L. and Blyxa aubertii Rich due to their presence in sampling site number S4 in the pre-monsoon and winter seasons. Similarly, joining of clustering was observed between Azolla filiculoides Lam. and Marsilea quadrifolia L. due to their presence in the winter seasons of all the sampling sites. Cyperus corymbosus Rottb., Cyperus rotundus L., and Panicum sp. joined clustering due to their presence in the pre-monsoon and winter seasons of all their sampling sites, Carex sp., Cyperus bulbosus Vahl and Cyperus compressus L. joined clustering due to their presence in S1, S2 and S3 sampling sites in pre-monsoon and winter seasons, Sagittaria guayanensis Kunth, Colocasia esculenta (L.) Schott, Hydrilla sp., Utricularia aurea Lour. Limnophila heterophylla (Roxb.) Benth., and Potamogeton nodosus Poir. have joined clustering due to their presence in pre-monsoon, retreating monsoon and winter seasons of all the sampling sites, Commelina benghalensis L., Axonopus compressus (Sw.) P. Beauv., Monochoria vaginialis (Burm.f.) C. Presl, Echinochloa sp., Eragrostis sp. and Ludwigia adscendens (L.) H. Hara has joined clustering due to their presence in the pre-monsoon and winter seasons at S1 and S2 sampling sites. In the dendrogram, Eichhornia crassipes (Mart.) Solms. was identified as an out-group, without joining clustering due to its presence in the monsoon and retreating monsoon seasons in all the sampling sites. Similarly, Lemna perpusilla Torr was also identified as an out-group due to its presence in the retreating monsoon and winter season of S1 and S2 sampling sites. All other macrophytes have joined clustering due to their presence in the pre-monsoon, retreating monsoon and winter seasons at the sampling sites number S1 and S2. In addition to the above-mentioned species of macrophytes, the unbranched filamentous freshwater green algae Spirogyra (Chlorophyta), of family zygnemataceae, forms free-floating mats in shallow waters during the retreating monsoon, winter and pre-monsoon seasons in all the sampling sites.
Table 1.2: Analysis of the aquatic macrophyte species of the Naljhara River
| Group | Family | Genera | Species | ||
|---|---|---|---|---|---|
| Number | % | Number | % | ||
| Angiosperms | Alismataceae | 1 | 3.23 | 1 | 2.78 |
| Amaranthaceae | 1 | 3.23 | 2 | 5.56 | |
| Aponogetonaceae | 1 | 3.23 | 1 | 2.78 | |
| Araceae | 1 | 3.23 | 1 | 2.78 | |
| Asteraceae | 1 | 3.23 | 1 | 2.78 | |
| Ceratophyllaceae | 1 | 3.23 | 1 | 2.78 | |
| Commelinaceae | 1 | 3.23 | 1 | 2.78 | |
| Cyperaceae | 2 | 6.45 | 5 | 13.89 | |
| Hydrocharitaceae | 3 | 9.68 | 3 | 8.33 | |
| Lemnaceae | 1 | 3.23 | 1 | 2.78 | |
| Lentibulariaceae | 1 | 3.23 | 1 | 2.78 | |
| Menyanthaceae | 1 | 3.23 | 1 | 2.78 | |
| Onagraceae | 1 | 3.23 | 1 | 2.78 | |
| Plantaginaceae | 1 | 3.23 | 1 | 2.78 | |
| Poaceae | 7 | 22.58 | 7 | 19.44 | |
| Polygonaceae | 1 | 3.23 | 1 | 2.78 | |
| Pontederiaceae | 2 | 6.45 | 2 | 5.56 | |
| Potamogetonaceae | 1 | 3.23 | 2 | 5.56 | |
| Typhaceae | 1 | 3.23 | 1 | 2.78 | |
| Pteridophytes | Azollaceae | 1 | 3.23 | 1 | 2.78 |
| Marsileaceae | 1 | 3.23 | 1 | 2.78 | |
| Total = 21 | 31 | 36 | |||
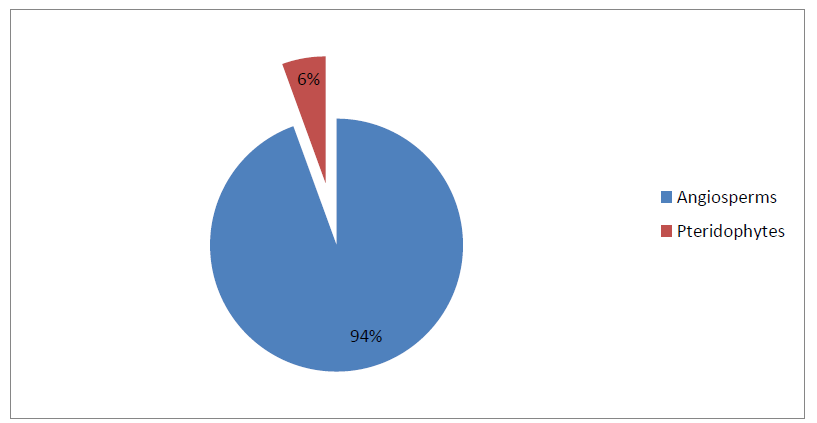
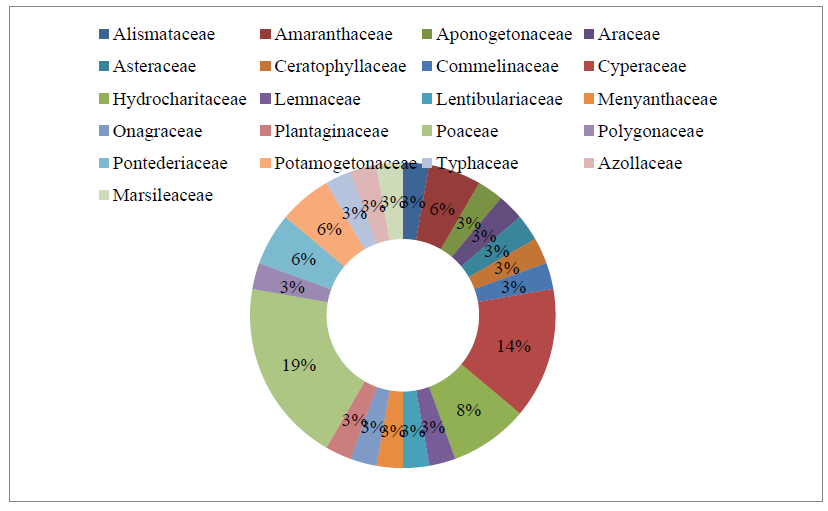
Table 1.3: Ecological categorization of Macrophyte species of the Naljhara river
| Sl.No. | Categories | Name of species | No. of species | % of species |
|---|---|---|---|---|
| 1 | Free floating |
Azolla filiculoides Lam. Eichhornia crassipes (Mart.) Solms. Lemna perpusilla Torr. Utricularia aurea Lour. |
4 | 11.11 |
| 2 | Rooted emergent with floating leaves |
Aponogeton sp. Hydrocharis sp. Potamogeton nodosus Poir. Sagittaria guayanensis Kunth |
4 | 11.11 |
| 3 | Rooted submerged |
Blyxa aubertii Rich Ceratophyllum sp. Hydrilla sp. Potamogeton crispus L. |
4 | 11.11 |
| 4 | Rooted emergent or marginal |
Alternanthera philoxeroides (Mart.) Alternanthera sessilis (L.) R.Br. ex DC. Axonopus compressus (Sw.) P. Beauv. Carex sp. Colocasia esculenta (L.) Schott. Commelina benghalensis L. Cyperus bulbosus Vahl. Cyperus compressus L. Cyperus corymbosus Rottb. Cyperus rotundus L. Digitaria sp. Echinochloa sp. Enhydra fluctuans Lour. Eragrostis sp. Hygoryza aristata (Retz.) Nees ex Wight & Arn. Limnophila heterophylla (Roxb.) Benth. Limnanthmum sp. Ludwigia adscendens (L.) H. Hara Marsilea quadrifolia L. Monochoria vaginialis (Burm.f.) C. Presl. Panicum sp. Paspalum sp. Polygonum plebeium R.Br. Typha sp. |
24 | 66.67 |
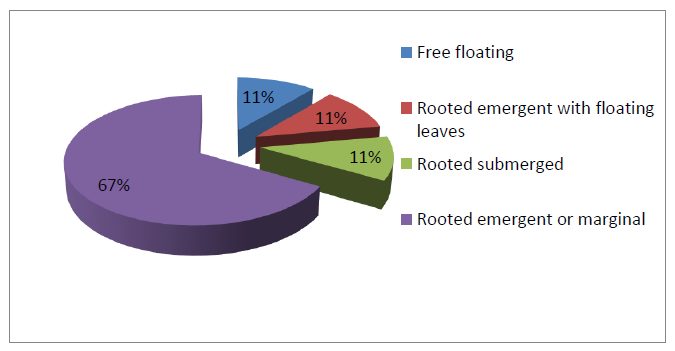
Table 1.4: Distribution status of aquatic macrophytes recorded in different seasons during the year 2022-2023 in Naljhara River in four sampling sites (PM = Pre-monsoon; M= Monsoon; RM= Retreating monsoon; W= winter; S1= Sampling site 1; S2= Sampling site 2; S3= Sampling site 3; S4= Sampling site 4)
| Group | Family | Names of Species | S1 | S2 | S3 | S4 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| PM | M | RM | W | PM | M | RM | W | PM | M | RM | W | PM | M | RM | W | |||
|
Angiosperm
|
Alismataceae | Sagittaria guayanensis Kunth |
+ | - | + | + | + | - | + | + | + | - | + | + | + | - | + | + |
| Amaranthaceae | Alternanthera sessilis (L.) R.Br. ex DC. |
+ | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Alternanthera philoxeroides (Mart.) Griseb |
+ | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | ||
| Aponogetonaceae | Aponogeton sp | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Araceae | Colocasia esculenta (L.) Schott. |
+ | - | + | + | + | - | + | + | + | - | + | + | + | - | + | + | |
| Asteraceae | Enhydra fluctuansLour. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Ceratophyllaceae | Ceratophyllum sp. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Commelinaceae | Commelina benghalensisL. |
+ | - | - | + | + | - | - | + | - | - | - | - | - | - | - | - | |
| Cyperaceae | Carex sp. | + | - | - | + | + | - | - | + | + | - | - | + | - | - | - | - | |
| Cyperus bulbosus Vahl. | + | - | - | + | + | - | - | + | + | - | - | + | - | - | - | - | ||
| Cyperus compressus L. | + | - | - | + | + | - | - | + | + | - | - | + | - | - | - | - | ||
| Cyperus corymbosus Rottb. | + | - | - | + | + | - | - | + | + | - | - | + | + | - | - | + | ||
| Cyperus rotundus L. | + | - | - | + | + | - | - | + | + | - | - | + | + | - | - | + | ||
| Hydrocharitaceae | Blyxa aubertii Rich. | - | - | - | - | - | - | - | - | + | - | - | + | + | - | - | + | |
| Hydrilla sp. | + | - | + | + | + | - | + | + | + | - | + | + | + | - | + | + | ||
| Hydrocharis sp. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | ||
| Lemnaceae | Lemna perpusilla Torr. | - | - | + | + | - | - | + | + | - | - | - | - | - | - | - | - | |
| Lentibulariaceae | Utricularia aurea Lour. | + | - | + | + | + | - | + | + | - | - | + | + | + | - | + | ||
| Menyanthaceae | Limnanthmum sp. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Onagraceae | Ludwigia adscendens (L.) H. Hara |
+ | - | - | + | + | - | - | + | - | - | - | - | - | - | - | - | |
| Plantaginaceae | Limnophila heterophylla (Roxb.) Benth. |
+ | - | + | + | + | - | + | + | + | - | + | + | + | - | + | + | |
| Poaceae | Axonopus compressus (Sw.) P. Beauv. |
+ | - | - | + | + | - | - | + | - | - | - | - | - | - | - | - | |
| Digitaria sp. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | ||
| Echinochloa sp. | + | - | - | + | + | - | - | + | - | - | - | - | - | - | - | - | ||
| Eragrostis sp. | + | - | - | + | + | - | - | + | - | - | - | - | - | - | - | - | ||
| Hygoryza aristata (Retz.) Nees ex Wight & Arn. |
+ | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | ||
| Panicum sp. | + | - | - | + | + | - | - | + | + | - | - | + | + | - | - | + | ||
| Paspalum sp. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | ||
| Polygonaceae | Polygonum plebeium. R.Br. |
+ | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Pontederiaceae | Eichhornia crassipes (Mart.) Solms. |
- | + | + | - | - | + | + | - | - | + | + | - | - | + | + | - | |
| Monochoria vaginialis (Burm.f.) C. Presl. |
+ | - | - | + | + | - | - | + | - | - | - | - | - | - | - | - | ||
| Potamogetonaceae | Potamogeton nodosus Poir. |
+ | - | + | + | + | - | + | + | + | - | + | + | + | - | + | + | |
| Potamogeton crispus L. | - | - | - | - | - | - | - | - | - | - | - | - | + | - | - | + | ||
| Typhaceae | Typha latifolia L. | + | - | + | + | + | - | + | + | - | - | - | - | - | - | - | - | |
| Pteridophytes | Azollaceae | Azolla filiculoides Lam. | - | - | - | + | - | - | - | + | - | - | - | + | - | - | - | + |
| Marsileaceae | Marsilea quadrifolia L. | - | - | - | + | - | - | - | + | - | - | - | + | - | - | - | + | |
Note: “+” Symbol indicates present and “-” Symbol indicates absent.
Table 1.5: Number of species of aquatic macrophytes observed during the study period in the Naljhara river in different seasons and at selected sampling sites (PM = Pre-monsoon; M= Monsoon; RM= Retreating monsoon; W= winter; S1= Sampling site 1; S2= Sampling site 2; S3= Sampling site 3; S4= Sampling site 4)
| Sampling site with seasons | S1 PM |
S1 M |
S1 RM |
S1 W |
S2 PM |
S2 M |
S2 RM |
S2 W |
S3 PM |
S3 M |
S3 RM |
S3 W |
S4 PM |
S4 M |
S4 RM |
S4 W |
| Number of Species | 30 | 1 | 21 | 33 | 30 | 1 | 20 | 33 | 13 | 1 | 7 | 15 | 11 | 1 | 7 | 13 |

Conclusion
The present study demonstrated that in spite of widespread anthropogenic effects witnessed by different freshwater ecosystems across the globe, the river Naljhara, being a part of the Manas-Beki river system, still has rich macrophyte diversity. The composition of the aquatic macrophytes of the river demonstrated in the present study is a testimony to the existence of a healthy freshwater ecosystem. Although macrophyte composition and abundance give the status of an aquatic ecosystem, and any change can indicate the variation in the freshwater ecosystem, due to low seasonal variability, they exhibit slow changes in community structure. Thus, macrophytes have limitations in use as an early warning indicator to detect changes in the riverine ecosystem. The present study demonstrated a rich status of macrophyte species composition in the river Naljhara, especially during the pre-monsoon, retreating monsoon and winter seasons in all sampling sites. It is due to low water depth and flow during such seasons. Uses of other sensitive biotic indicators with macrophytes have the potential to determine the status of the health of this freshwater ecosystem.
Acknowledgement: The authors’ thanks are due to the Department of Botany B. H. College, Howly, for providing the necessary support during different phases of the study.
Declaration
Conflict of Interest: The authors declare that they have no conflict of interest.
References
- Biswas, K., and Calder, C.C. (1984). Handbook of common water and marsh plants of India and Burma. XVI +216. Dehra Dun: Bishen Singh Mahendra Pal Singh.
- Borthakur, M. (1986). Weather and climate in N. E. India. Northeastern Geographer 18 (1 & 2): 20-27.
- Campbell, J. M., Clark, W. J., & Kosinski, R. (1982). A technique for examining the microspatial distribution of Cladocera associated with shallow water macrophytes. Hydrobiologia, 97, 225–232.
- Chanu, M., Saikia, A. P., Pratim, P., Bairagi, A. K., & Bhagawati, P. (2022). Diversity of Aquatic Macrophytes of Borbila and Silsako Wetland in Assam. Research Journal of Agricultural Sciences, 13(5), 1339-1344.
- Dey, S.C. & Kar, D. (1989). Aquatic macrophytes of Lake Sone in Assam. Environment & Ecology 7(1) 253–254.
- Dey, S.C. (1981). Studies on the hydrobiological conditions of some commercially important lakes (Beels) of Kamrup district of Assam and their bearing on fish production. Final Technical Report, North-Eastern Council, Govt. of India 177.
- Diehl, S. (1992). Fish predation and benthic community structure—The role of omnivory and habitat complexity. Ecology, 73, 1646–1661.
- Dutta Ranjit and Sarma Sarada Kanta. (2012). Lower Subansiri Hydroelectric Power Project and the future of the Subansiri River Ecosystem. Annals of Biological Research, 3(6), 2953–2957.
- Fassett, N. C. (1997). A Manual of Aquatic Plants. Allied Scientific Publishers, Bikaner, India, Pp. 1–382.
- Hazarika, S. and Borthakur, S. K. (2014). Hydrophytic flora of Assam: II. Diversity of aquatic and wetland vascular plants of Nagaon District of Assam, India. Pleione 8(1) 96 - 108.
- Jain, S. K., and Rao, R. R. (1977). Field and Herbarium technique. Todays and Tomorrow Publ., New Delhi.
- Johnson, R. K. (2001). Indicator metrics and detection of impact. In e. K. Karttunen (Ed), In Monitoring and assessment of ecological status of aquatic environments. (pp. 41-44). TemaNord 563, Nordic Council of Ministers.
- Kar, D., & Barbhuiya, M. H. (2001). Ecology of aquatic macrophytes of Chatla Haor, a floodplain wetland in Cachar district of Assam. Environment & Ecology 19, 231–233.
- Lodge, D. M. (1991). Herbivory on freshwater macrophytes. Aquatic Botany, 41, 195–224.
- NERC. (1999). The UK Environmental Change Network Protocols for Standard Measurements at Freshwater Sites. www.ecn.ac.uk .
- Pradhan, P., Mishra, S. S., Chakraborty, S. K., & Bhakat, R. K. (2005). Diversity of freshwater macrophyte vegetation of six rivers of south West Bengal. Tropical Ecology, 46(2), 193–202.
- Rennie, M. D., & Jackson, L. J. (2005). The influence of habitat complexity on littoral invertebrate distributions: Patterns differ in shallow prairie lakes with and without fish. Canadian Journal of Fisheries and Aquatic Science, 62, 2088–2099.
- Riis, T., Tank, J. L., Reisinger, A. J., Aubena, A., Roche, K. R., Levi, P. S., … & Bolster, D. (2020). Riverine macrophytes control seasonal nutrient uptake via both physical and biological pathways. Freshwater Biology, 65(2), 178–192.
- Rulík, M., Opatřilová, L., Jurajda, P., Špaček, J., & Grulich, V. (2019). Rivers in the Czech Republic. In Assessment and Protection of Water Resources in the Czech Republic (pp. 39-69). Cham: Springer International Publishing.
- Sarma, S. K., & Deka, U. (2014). Quantitative analysis of macrophytes and physico-chemical properties of water of two wetlands of Nalbari district of Assam, India. Annals of Biological Research, 5(5), 77-84.
- Selthrope, C. D. (1985). The Biology of Aquatic vascular plants. Königstein-West Germany: Koeltz Scientific Books, Germany.
- Shah, J. D., & Abbas, S. G. (1979). Seasonal variation in frequency, density, biomass and rate of production of aquatic macrophytes of the River Ganga at Bhagalpur, Bihar. Tropical Ecology, 20, 127-134.
- UNEP. (2004). Integrated watershed management-Ecohydrology and phytotechnology-Manual. Osaka, Japan: United Nations publication.
- Wetzel, R. G., & Allen, H. L. (1970). Functions and interaction of dissolved organic matter and the littoral zone in lake metabolism and eutrophication. In e. In Z. Kajak and A. Hillbrichtilkowska (Ed), Productivity Problems of Freshwaters. (pp. 333-347). Warsaw: PWN Polish scientific publishers.
- Williams, D. D. (1980). Some relationships between stream benthos and substrate heterogeneity. Limnology and Oceanography, 25, 161–172.
- Wilzbach, M. A. (1985). Relative roles of food abundance and cover in determining the habitat distribution of streamdwelling cutthroat trout (Salmo clarki). Canadian Journal of Fisheries and Aquatic Sciences, 42, 1668–1672.